The Food and Drug Administration issued the world’s first approval of a vaccine against respiratory syncytial virus (RSV), an achievement that researchers have worked toward for decades after a catastrophic clinical trial in the 1960s.
The vaccine, called Arexvy from pharmaceutical giant GSK, is approved for adults aged 60 and over. The Centers for Disease Control and Prevention will need to recommend the vaccine before it will be available for use. The agency’s advisory committee for immunizations is next scheduled to meet June 21 to 22 and could discuss the vaccine then.
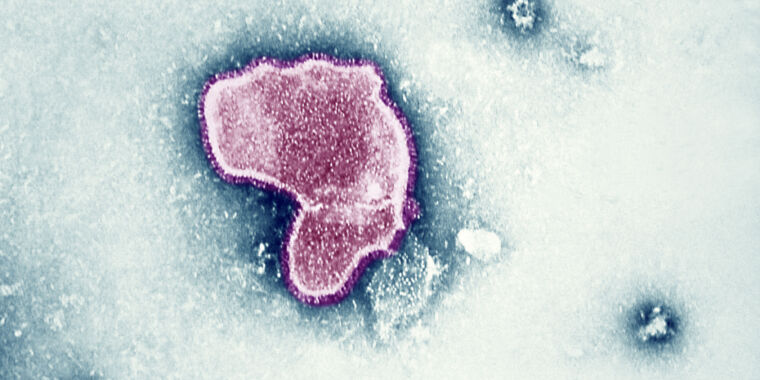
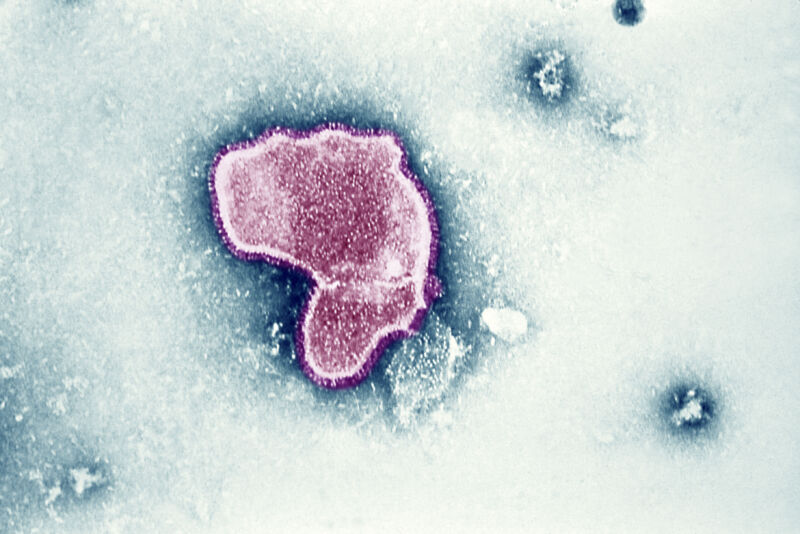
RSV is a common, highly contagious seasonal respiratory infection. It’s often associated with infants, who are at especially high risk of developing severe disease. In fact, bronchitis from RSV infections is the leading cause of hospitalization among infants under the age of 1 in the US. But the virus is also dangerous for older adults, causing an estimated 60,000 to 160,000 hospitalizations and 6,000 to 10,000 deaths in older adults each year in the country.
In an ongoing, randomized, placebo-controlled clinical study of adults aged 60 and older—in which about 12,500 participants received Arexvy, and 12,500 participants received a placebo— the vaccine reduced the risk of developing lower-respiratory tract infection from RSV by 82.6 percent, and reduced the risk of developing a severe RSV infection by 94.1 percent, the FDA noted.
“Older adults, in particular those with underlying health conditions, such as heart or lung disease or weakened immune systems, are at high risk for severe disease caused by RSV,” Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said in a statement. The approval Wednesday of this first RSV vaccine is “an important public health achievement to prevent a disease which can be life-threatening,” he added.
Triumph after tragedy
GSK also celebrated the landmark approval, with GSK’s Chief Scientific Officer Tony Wood saying the approval “marks a turning point in our effort to reduce the significant burden of RSV.”
GSK’s vaccine is just the first of several RSV vaccines in the works. Pfizer has an RSV vaccine for adults 60 and up that was nearly 86 percent effective against severe disease in a phase III trial. It is now being reviewed by the FDA and is expected to gain approval this month. In January, Moderna said its RSV vaccine for older adults was 82 percent effective against severe disease.
Pfizer also has a vaccine to protect infants that is nearing a regulatory decision. When given to pregnant participants in a phase III trial, the vaccine was 82 percent effective at preventing severe RSV in the infant’s first three months and 69 percent effective over the first six months.
The vaccines are a triumph after researchers spent decades working to find a safe and effective design for the immunizations. Small trials of early vaccine candidates in the 1960s led to children developing more severe RSV than unvaccinated children. Two vaccinated children died following their infections.
Subsequent research showed that those early, doomed vaccines presented a key protein—the F protein—in the wrong conformation, spurring the immune system to generate impotent antibodies and exaggerated inflammatory responses that made the disease worse. It wasn’t until decades later that researchers at the National Institutes of Health figured out how to lock the F protein into a conformation that would induce an effective immune response, paving the way for the current vaccine designs.