After years of delays, regulatory rejections, and allegations of animal abuse, Elon Musk’s brain-computer interface company, Neuralink, is now recruiting its first human volunteers to have an experimental robot implant an experimental device directly into their brains.
In a blog post Tuesday, the company announced that an independent institutional review board and an unnamed hospital site granted approval for the trial to start recruiting volunteers.
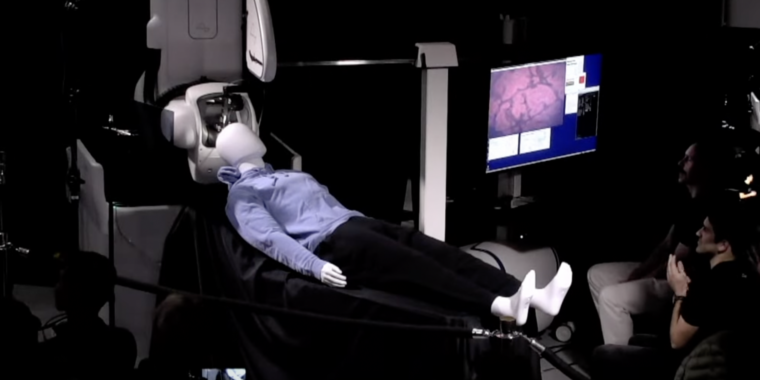
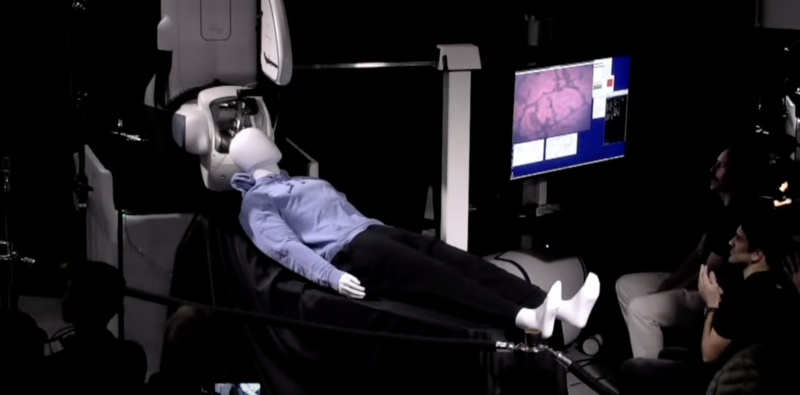
Neuralink says it aims to enroll people with quadriplegia due to a spinal cord injury or ALS (amyotrophic lateral sclerosis). Volunteers will have a wireless brain-computer interface implant, dubbed N1, surgically embedded into their brains by the company’s experimental surgical robot, R1. The implant device is said to have 1,024 electrodes distributed across 64 threads thinner than a human hair. After R1 inserts the threads into the appropriate brain region, the electrodes are designed to record neural activity related to movement intention, and an experimental app from the company will decode the signals. The goal of the N1 implantation is to allow trial participants to control a computer cursor or keyboard using only their thoughts. This trial will primarily evaluate safety, but also get a glimpse of efficacy, Neuralink says.
There’s little information about the trial beyond what’s included in Neuralink’s blog and its two-page study brochure for prospective volunteers. The clinical trial still does not appear to be registered on the federal database for clinical trials (clinicaltrails.gov). Multiple searches for trials matching the description of Neuralink’s trial returned no corresponding results. Most notably, it remains unclear how many people Neuralink will try to recruit for this initial clinical trial. The company had previously tried to get approval from the Food and Drug Administration to recruit 10 people. But, according to reporting from Reuters, Neuralink tried negotiating a lower number after the FDA raised safety concerns.
The trial is overdue based on Musk’s timelines and the progress of rival brain chip companies. In a July 2019 presentation, the billionaire said the company, founded in 2016, aimed to have regulatory approval for a human trial by the end of 2020. But, according to current and former FDA officials who spoke with Reuters, Neuralink didn’t seek permission until early 2022—and then the FDA rejected its application. The rejection was based on several issues and safety concerns, including the reliability and safety of the device’s lithium batteries, the possibility of migration of the device’s threads, and uncertainty as to whether the device can be removed without damaging brain tissue.
Meanwhile, interviews with current and former Neuralink employees revealed allegations that the company had abused research animals, including performing “hack job” surgeries and euthanizing more animals than necessary. Some employees blamed the sloppy errors and abuse on Musk’s rushed timelines and unrealistic demands. In 2022, he reportedly sent an email to employees about a rival company making progress with an implant that enabled a paralyzed man to walk, then followed the message up with another that read: “In general, we are simply not moving fast enough. It is driving me nuts!”
In May, Neuralink announced it had received the green light from the FDA to start a human trial. The trial—called the PRIME study for Precise Robotically Implanted Brain-Computer Interface—will take over six years to fully complete. Participants will primarily be evaluated in nine at-home and in-clinic visits over 18 months. Then they will transition to a long-term follow-up part of the study, which will include 20 visits over five years.