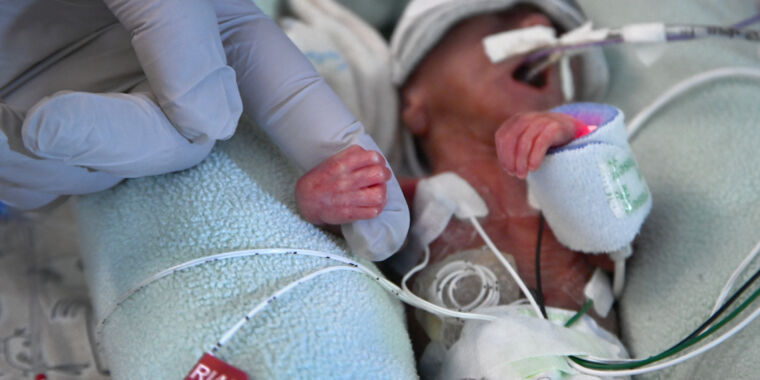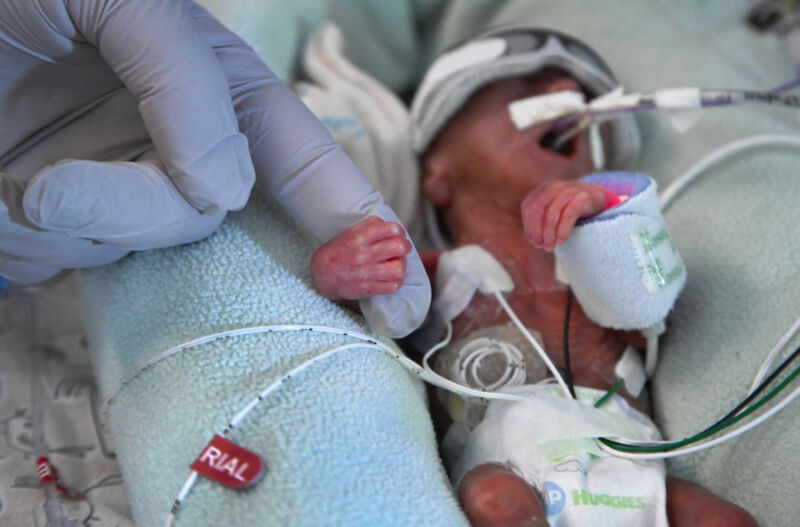
The Food and Drug Administration is warning health care providers not to use probiotics containing live bacteria or yeast in preterm infants after the agency began investigating the July death of a preterm, low-weight infant given such a product in an unnamed hospital.
The infant developed sepsis from the bacterium in the probiotic product—Evivo with MCT Oil made by Infinant Health—and subsequently died.
In a statement to Ars, the FDA said it quickly investigated the death after receiving an initial report on July 31. “Infant deaths are especially tragic and determining causality of preterm infant death can be particularly complicated,” an agency spokesperson said. The agency reviewed medical records and laboratory tests from the case and collected clinical samples and product samples for analysis.
“In September 2023, genomic sequencing data analysis performed by the FDA found that the probiotic bacteria present in Evivo with MCT Oil was a genetic match to the bacterium isolated from the infant’s blood,” the agency said.
The bacteria in the product and found in the infant was Bifidobacterium longum subspecies infantis.
The FDA did not respond to Ars’ question about the state or hospital where the death occurred.
On Friday, the FDA sent out its warning to health care providers about the dangers of probiotics in infants, particularly preterm infants that are highly vulnerable. The agency noted that the case isn’t the first of its kind; there have been previous reports of infections and sepsis in infants due to the use of probiotics containing bacteria and yeast.
“The FDA is also reminding healthcare providers that FDA has not approved any probiotic product for use as a drug or biological product in infants,” the agency wrote in the warning.
Unproven, risky treatment
Probiotics are generally not recommended for infants, particularly vulnerable ones. The American Academy of Pediatrics recommends against them. “Given the lack of FDA-regulated pharmaceutical-grade products in the United States, conflicting data on safety and efficacy, and potential for harm in a highly vulnerable population, current evidence does not support the routine, universal administration of probiotics to preterm infants, particularly those with a birth weight of <1000 g.” Still, a 2021 report found that approximately 10 percent of very premature infants in the US receive some sort of probiotic preparation while receiving specialized care in hospitals.
While the FDA tried to inform health care providers about the dangers of probiotics, the agency also sent a rebuking letter to the maker of Evivo with MCT Oil: Infinant Health, Inc., formerly Evolve Biosystems Inc. The agency criticized the California-based company for clearly marketing its unapproved product—sold as a dietary supplement—for use in the prevention of a serious disease in highly vulnerable pre-term infants. Moreover, the agency took aim at the company touting the supplement as “made specifically for use in healthcare settings,” and “[d]esigned specifically for infants in the NICU[.]” NICU is the Neonatal Intensive Care Unit, where the most vulnerable of newborns, many preterm, receive specialized and intensive care.