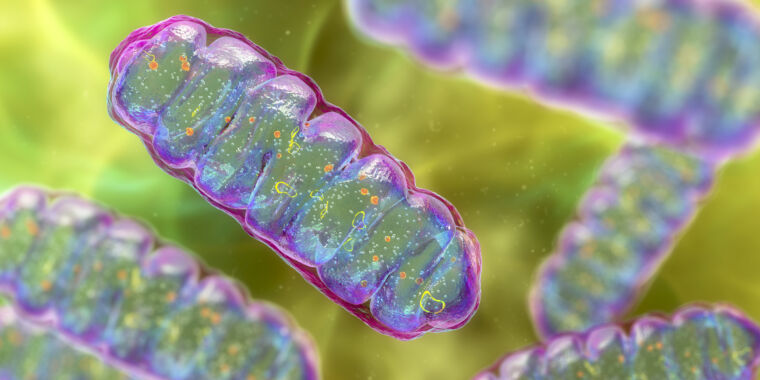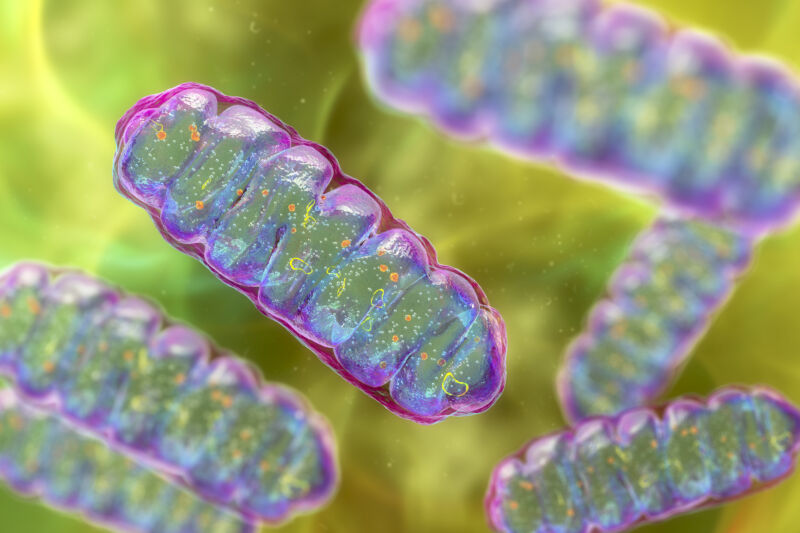
More than 1.5 billion years ago, a momentous thing happened: Two small, primitive cells became one. Perhaps more than any event—barring the origin of life itself—this merger radically changed the course of evolution on our planet.
One cell ended up inside the other and evolved into a structure that schoolkids learn to refer to as the “powerhouse of the cell”: the mitochondrion. This new structure provided a tremendous energetic advantage to its host—a precondition for the later evolution of complex, multicellular life.
But that’s only part of the story. The mitochondrion is not the only important structure within complex, eukaryotic cells. There’s the membrane-bound nucleus, safekeeper of the genome. There’s a whole system of internal membranes: the endoplasmic reticulum, the Golgi apparatus, lysosomes, peroxisomes and vacuoles—essential for making, transporting, and recycling proteins and other cargo in and around the cell.
Where did all these structures come from? With events lost in the deep past and few traces to serve as evolutionary clues, it’s a very tough question to tackle. Researchers have proposed various hypotheses, but it is only recently, with some new tools and techniques, that cell biologists have been able to investigate the beginnings of this intricate architecture and shed some light on its possible origins.
A microbial merger
The idea that eukaryotes originated from two cells merging dates back more than 100 years but did not become accepted or well-known until the 1960s, when the late evolutionary biologist Lynn Margulis articulated her theory of endosymbiosis. The mitochondrion, Margulis said, likely originated from a class of microbes known as alphaproteobacteria, a diverse group that today includes the bacterium responsible for typhus and another one important for the genetic engineering of plants, among many others.
Nothing was known about the nature of the original host cell. Scientists proposed that it already was fairly complicated, with a variety of membrane structures inside it. Such a cell would have been capable of engulfing and ingesting things—a complicated and energetically expensive eukaryotic feature called phagocytosis. That might be how the mitochondrion first got into the host.
But this idea, called the “mitochondria late” hypothesis, doesn’t explain how or why the host cell had become complex to begin with.
In 2016, evolutionary biologist Bill Martin, cell biologist Sven Gould and bioinformatician Sriram Garg, at the University of Dusseldorf in Germany, proposed a very different model known as the “mitochondria early” hypothesis. They argued that since no primitive cells today have any internal membrane structures, it seems very unlikely that a cell would have had these over 1.5 billion years ago.
Instead, the scientists reasoned, the endomembrane system—the whole hodgepodge of parts found inside complex cells today — could have evolved soon after the alphaproteobacterium took up residence inside a relatively simple host cell, of a kind from a class called archaea. The membrane structures would have arisen from bubbles, or vesicles, released by the mitochondrial ancestor.
Free-living bacteria shed vesicles all the time, for all sorts of reasons, Gould, Garg, and Martin note, so it seems reasonable to think they’d continue to do that when enclosed inside a host.
Eventually, these vesicles would have become specialized for the functions that membrane structures perform today inside eukaryotic cells. They would even fuse with the host cell’s membrane, helping to explain why the eukaryote plasma membrane contains lipids with bacterial features.
Vesicles could have served an important initial function, says biochemist Dave Speijer of the University of Amsterdam. The new endosymbiont would have generated plenty of poisonous chemicals called reactive oxygen species, by oxidizing fatty acids and burning them for energy. “These destroy everything, they are toxic, especially on the inside of a cell,” Speijer says. Sequestering them inside vesicles would have helped keep the cell safe from harm, he says.
Another problem created by the new guest could also have been helped by making membrane barriers, Gould, Garg, and Martin add. After the alphaproteobacterium arrived, bits of its DNA would have mixed with the genome of the archaeal host, interrupting important genes. Fixing this would mean evolving machinery to splice out these foreign pieces—today they’re known as introns—from the messenger RNA copies of genes, so those protein-making instructions wouldn’t be garbled.
But that created yet another problem. The protein-making machinery—the ribosome—works extremely fast, joining several amino acids together per second. In contrast, the intron-removing system of the cell is slow, snipping out about one intron per minute. So unless the cell could keep the mRNA away from ribosomes until the mRNA was properly processed, the cell would produce many nonsensical, useless proteins.
The membrane surrounding the nucleus provided an answer. Serving as a spatial barrier, it allows mRNA splicing to finish up in the nucleus before the intron-free mRNA is translated in the cell’s internal fluid, the cytosol. “This is the selective pressure behind the origin of the nucleus,” Martin says. To form it, vesicles secreted by the endosymbiont would have flattened and wrapped around the genome, creating a barrier to keep ribosomes out but still allowing small molecules to pass freely.